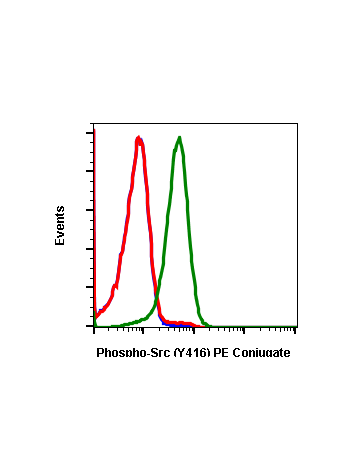
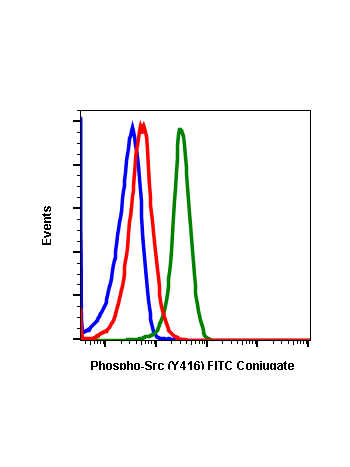
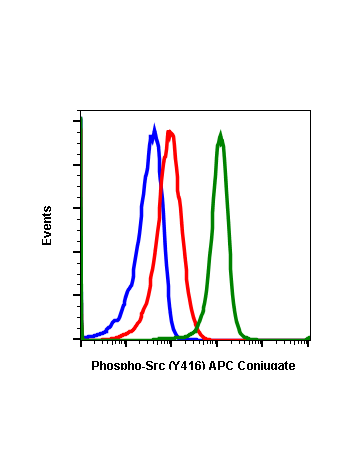
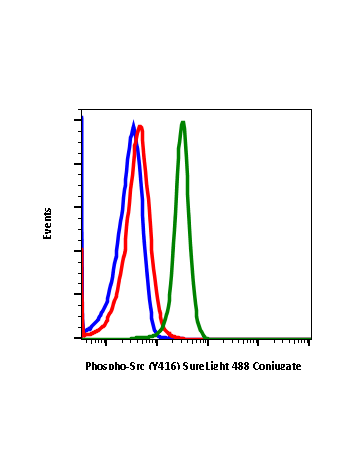
Phospho-Src (Tyr416) (C4) rabbit mAb
From
$210.00
In stock
Only %1 left
SKU
2071
Proto-oncogene tyrosine-protein kinase Src (c-Src, Src), is a non-receptor tyrosine kinase. Src phosphorylates specific tyrosine residues in other proteins and is involved in signal transduction and is important in human tumor development and poor prognosis in cancers. There are nine enzymes (Fyn, Yes, Hck, Lck, Lyn, Blk, Frk, Yrk, and Fgr) closely-related to Src and are collectively called the Src family kinases (SFKs)(1). SFKs represent the largest family of nonreceptor tyrosine kinases and interact directly with receptor tyrosine kinases, G-protein-coupled receptors (GPCRs), steroid receptors, signals transducers, and activators of transcription and molecules involved in cell adhesion and migration. These events lead to proliferation, cell growth, differentiation, cell shape, motility, migration, angiogenesis, and survival. Phosphorylation at Tyr416 in the activation loop of the kinase domain upregulates phospho Src activity, while phosphorylation at Tyr527 in the carboxy-terminal tail decreases activity. Src has been strongly implicated in the development, maintenance, progression, and metastatic spread of several human cancers such as prostate, lung, breast, and colorectal. Structurally, SFKs are highly related to one another and contain conserved structural elements between family members. These elements include the N-terminal Src Homology 4 domain (SH4), the Src Homology 3 domain (SH3), Src Homology 2 domain (SH2), a linker sequence, the tyrosine kinase domain, and the C-terminal tail (2). The N-terminal domain, SH4, serves as a site for myristoylation and targets SFKs to the cytoplasmic membrane. The SH3 domain binds amino acid sequences rich in proline residues (3). This domain is critical for Src activity, intracellular localization, and the recruitment and binding of Src substrates. The SH2 domain binds to short motifs containing phospho-tyrosines. Together, the SH2 and SH3 domains cooperate in regulating SFKs catalytic activity.
| Applications | Flow Cytometry |
|---|---|
| Clone | SrcY416-C4 |
| Format | Unconjugated |
| Validated Reactivity | Human, Mouse |
| Cross Reactivity | Predicted to work with mouse, rat and other homologues. |
| Detection | Anti-Rabbit IgG |
| Clonality | Monoclonal |
| Immunogen | A synthetic phospho-peptide corresponding to residues surrounding Tyr416 of human phospho Src |
| Formulation | 1X PBS, 0.02% NaN3, 50% Glycerol, 0.1% BSA |
| Isotype | Rabbit IgGk |
| Preparation | Protein A+G |
| Recommended Usage | 1µg/mL – 0.001µg/mL. It is recommended that the reagent be titrated for optimal performance for each application. See product image legends for additional information. |
| Storage | -20ºC |
| Pseudonyms | Proto-oncogene c-Src, pp60c-src, p60-Src, SRC1 |
| Uniprot ID | P12931 |
| References | (1) Src family kinases, key regulators of signal transduction. Parsons SJ, Parsons JT Oncogene. 2004 Oct 18; 23(48):7906-9. (2) Src protein-tyrosine kinase structure and regulation. Roskoski R Jr Biochem Biophys Res Commun. 2004 Nov 26; 324(4):1155-64. (3) Cohen GB, Ren R, Baltimore D. Modular binding domains in signal transduction proteins. Cell. 1995;80:237–248. |
Write Your Own Review