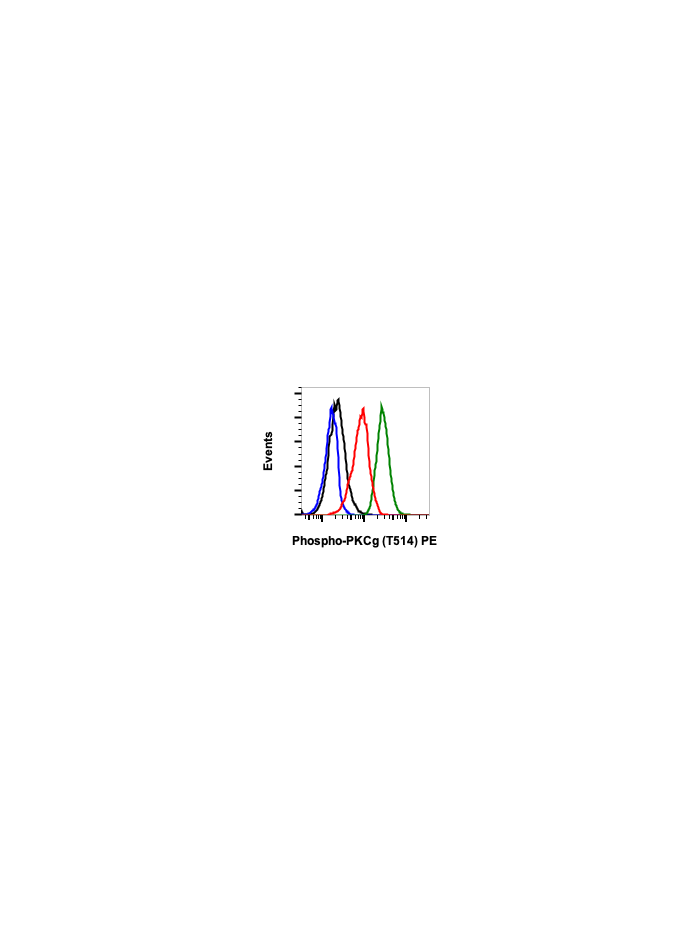
Phospho-PKC (pan) (gamma Thr514) (PF4) rabbit mAb PE conjugate
Protein Kinase (PKC) is a 12 member family of serine/threonine kinases termed conventional or classical (a b, g), novel (d,e,and q), atypical (z, l) and PKN and PKC-related (PKN1, PKN2 and PKN3) playing significant role in several signaling processes involved in physiological and pathological setting (1). PKC activation translates into gene expression modulation, cell division, migration, proliferation, differentiation, and cell survival and apoptosis (2). PKC members are classified based on their distinct cofactor requirements and the extent of homology between their regulatory elements (3,4). PKCalpha, beta I, beta II, and gamma constitute the conventional PKC isoforms, characterized by the presence of two cysteine-rich zinc finger domains, C1a and C1b (5,6) which bind to diacylglycerol (DAG) and phosphatidylserine (PS) (7). In addition, PKC contains a C2 domain, responsible for binding anionic phospholipids like phosphatidylinositol bisphosphate (PIP2) in a Ca2+-dependent manner (8,9). The atypical PKC, PKCz andl share ATP-binding domain and the catalytic domain with PKC. They contain a single C1 domain that lacks residues necessary for binding DAG (10).
Association of PKCg to the membrane enables a conformation that permits phosphoinositide-dependent protein kinase1 (PDK-1) (11) to bind and phosphorylate a site in the activation loop, Thr514. (12,13). Thr514 phosphorylation leads to a conformation change enabling phosphorylation of at two carboxyl-terminal sites namely, the turn motif and hydrophobic motif, as a result of which the fully phosphorylated conventional PKC is released from the membrane, and positioned in the cytoplasm as an inactive form (14,15). Binding of Ca2+ induces low-affinity interaction with the membrane, whereas the membrane imbedded cofactor DAG to PKC results in high-affinity interaction of PKC with the membrane (16).
| Applications | Flow Cytometry |
|---|---|
| Clone | PKCgT514-PF4 |
| Format | PE |
| Validated Reactivity | Human, Mouse, Rat |
| Cross Reactivity | Predicted to work with mouse, rat and other homologues. |
| Clonality | Monoclonal |
| Immunogen | A synthetic phospho-peptide corresponding to residues surrounding Thr514 of human phospho PKCγ |
| Formulation | 1X PBS, 0.09% NaN3, 0.2% BSA |
| Isotype | Rabbit IgGk |
| Preparation | Protein A+G |
| Recommended Usage | For flow cytometric staining, the suggested use of this reagent is 5 µL per million cells or 5 µL per 100 µL of staining volume. It is recommended that the reagent be titrated for optimal performance for each application. See product image legends for additional information. |
| Storage | 2-8ºC |
| Pseudonyms | Protein kinase C gamma type, PRKCG, PKCG |
| Uniprot ID | P17252 P05771 P05771-2 Q05655 Q02156 P24723 P05129 Q04759 |
| References | 1. Mugami S, et al., 2018, Mol. Cell. Endocrinol. 463:97-105. 2. Isakov N, 2018, Semin. Cancer Biol. 48:36-52. 3. Mellor H, and Parker PJ, 1998, Biochem. J. 332:281-92. 4. Newton AC, 1995, J. Biol. Chem. 270:28495-8. 5. Ho C, et al., 2001, Biochem. 40:10334-41. 6. Hurley JH, et al., 1996, Protein Sci. 6:477-80. 7. Colon-Gonzalez F, and Kazanietz MG, 2006, Biochim. Biophys. Acta 1761 :827-37. 8. Naleski EA, and Falke JJ, 1996, Protien Sci, 5 :2375-90. 9. Orr JW, and Newton AC, 1992, Biochem 31:4667-4673. 10. Antal CE and Newton AC, 2014, Biochem Soc. Trans. 42:1477-1483. 11. Sonnenberg ED, et al., 2001 J Biol Chem, 276:45289-97. 12. Dephoure N, et al., 2008, Proc. Natl. Acad Sci. U.S.A. 105:10762-67. 13. Barnett ME, et al., 2007, Cell Signal. 19:1820-9. 14. Newton AC, 2003, Biochem. J. 370 :361-71. 15. Parekh DB, et al., 2000, EMBO J. 10:496-503. 16. Edwards AS, et al., 1999, J Biol. Chem., 274:6461-8. |