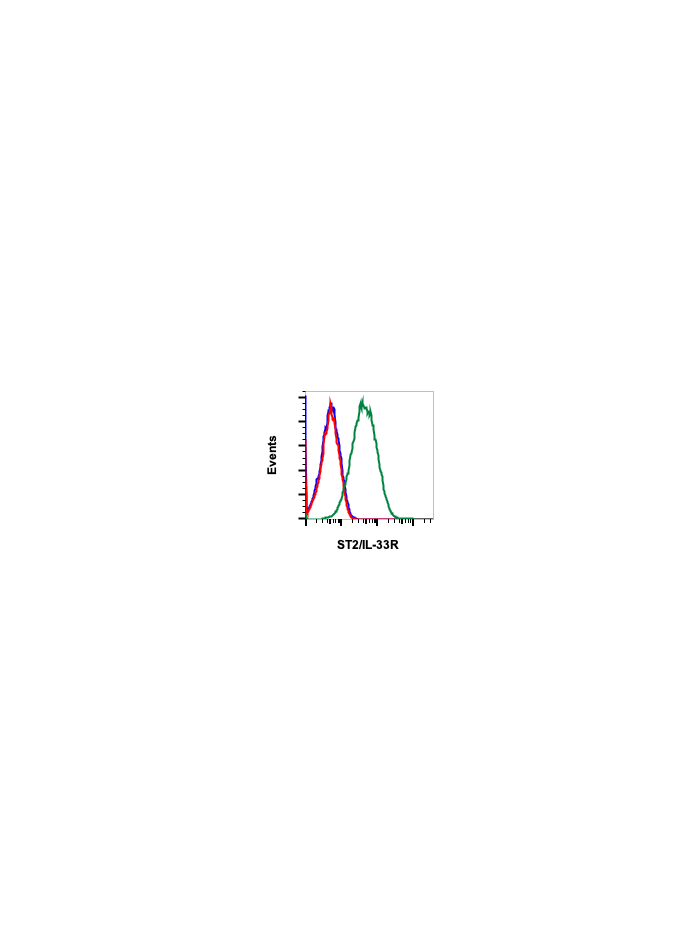
ST2/IL-33R (H2) rabbit mAb
Suppression of tumorigenicity (ST)2 is an interleukin (IL)-1 receptor family member and due to alternative splicing and 3’ processing at RNA level, ST2 is expressed as soluble (sST2) as well as trans-membrane (ST2L) isoforms (1). Originally identified in 1989 as an orphan receptor, the ST2 ligand was identified to be IL-33 in 2005 (2). IL-33 is mechanically induced in cardiac fibroblasts and antagonizes hypertrophic stimuli. Analysis of rat neonatal cardiac fibroblasts and cardiomyocytes indicate that gene expression of IL-33 and sST2 was more than 5-fold greater in cardiac fibroblasts than in cardiomyocytes (3,4). Expression sST2 is markedly increased as early as 1 hr following mechanical strain in cultured myocytes and in patients with acute myocardial infarction (5).
In addition, IL-33 behaves as a chromatin-related nuclear interleukin, and act as transcriptional repressor when overexpressed in cells (4). IL-33 production may be augmented by inflammation (6). IL-33 has been demonstrated to participate in several forms of inflammatory diseases and to influence tumorigenesis. IL-33 was identified to be pro-cancer mediator, since the activation pathway of IL-33/ST2 could promote metastasis (7).
| Applications | Flow Cytometry |
|---|---|
| Clone | ST2IL33R-H2 |
| Format | Unconjugated |
| Validated Reactivity | Human |
| Cross Reactivity | Predicted to work with mouse, rat and other homologues. |
| Detection | Anti-Rabbit IgG |
| Clonality | Monoclonal |
| Immunogen | human recombinant ST2-Fc |
| Formulation | 1X PBS, 0.09% NaN3, 0.2% BSA |
| Isotype | Rabbit IgGk |
| Preparation | Protein A+G |
| Recommended Usage | For flow cytometric staining, the suggested use of this reagent is 5 µL per million cells or 5 µL per 100 µL of staining volume. It is recommended that the reagent be titrated for optimal performance for each application. See product image legends for additional information. |
| Storage | 2-8ºC |
| Pseudonyms | Suppression Tumorigenicity (ST)2, IL-33 receptor, IL-1 receptor |
| Uniprot ID | Q01638 |
| References | 1. Schmitz J, et al., (2005) Immunity, 23: 479-490. 2. Dinarello CA, (2005), Immunity, 23: 461-462. 3. Weinberg EO, et al., (2003), Circulation, 107: 721-726 4. Sanada S. et al., (2007), J Clin Invest, 117: 1538-1549. 5. Shimpo M., et al., (2004), Circulation, 109: 2186-2190. 6. Kim S., al at., (2011), Ann N.Y. Acad. Sci., 1217:191-206. 7. Afferni C., et al., (2018), Front Immunol, 9: 2601. doi:10.3389/fimmu.2018.02601 |